Yersinia pestis antibiotic resistance: a systematic review
Article information
Abstract
Yersinia pestis, the cause of plague and a potential biological weapon, has always been a threatening pathogen. Some strains of Y. pestis have varying degrees of antibiotic resistance. Thus, this systematic review was conducted to alert clinicians to this pathogen’s potential antimicrobial resistance. A review of the literature was conducted for experimental reports and systematic reviews on the topics of plague, Y. pestis, and antibiotic resistance. From 1995 to 2021, 7 Y. pestis isolates with 4 antibiotic resistance mechanisms were reported. In Y. pestis 17/95, 16/95, and 2180H, resistance was mediated by transferable plasmids. Each plasmid contained resistance genes encoded within specific transposons. Strain 17/95 presented multiple drug resistance, since plasmid 1202 contained 10 resistance determinants. Strains 16/95 and 2180H showed single antibiotic resistance because both additional plasmids in these strains carried only 1 antimicrobial determinant. Strains 12/87, S19960127, 56/13, and 59/13 exhibited streptomycin resistance due to an rpsl gene mutation, a novel mechanism that was discovered recently. Y. pestis can acquire antibiotic resistance in nature not only via conjugative transfer of antimicrobial-resistant plasmids from other bacteria, but also by gene point mutations. Global surveillance should be strengthened to identify antibiotic-resistant Y. pestis strains by whole-genome sequencing and drug susceptibility testing.
Introduction
Yersinia pestis, an obligate parasite and Gram-negative bacterium, is the causative agent of plague and has killed an estimated 200 million people in 3 major pandemics in the 14th, 17th, and 19th centuries. The first pandemic, known as the Black Death, killed more than 30% of Europe’s population. Alexandre Yersin, a bacteriologist, discovered it during a plague investigation in Hong Kong in 1894 [1]. Plague is now rare, and it is generally considered as a disease of the past. However, it still appears in many areas, including Asia, Africa, and America, in the 21st century, and it remains a threat to public health [2]. Every year, at least 2,000 cases of plague are documented. Y. pestis can grow within a wide range of temperatures, and its optimal pH for growth is 7.2 to 7.6 [3]. It dies rapidly when exposed to ultraviolet light, intensive desiccation, temperatures exceeding 40°C [4].
It is generally accepted that Y. pestis evolved from Yersinia pseudotuberculosis 1,500–2,000 years ago [5]. However, Y. pestis was recently found in the remains of a hunter-gatherer from 5,000 years ago via ancient DNA analysis, suggesting that Y. pestis emerged about 7,000 years ago, which is substantially older than considered before [6]. This early strain isolated from ancient human remains seems to be less virulent and less contagious than later strains of Y. pestis [6]. Y. pestis strain CO92 was the first to be sequenced [7], and other strains have been sequenced subsequently [8–11]. Specific characteristics of the genome and molecular mechanisms of Y. pestis contribute to its infection of fleas, virulence, and subsistence against the host immune response [12].
According to biologists, many wild animal species are highly susceptible to Y. pestis. It lives in small rodents that are mostly found in rural and semi-rural areas of Africa, Asia, and the United States. The effects of plague on these populations are unknown, but certain characteristics make rodent species more vulnerable to the disease. However, due to a series of gene mutations, gene acquisition and loss, the molecular mechanisms and lifestyle of Y. pestis have changed dramatically [5]. Y. pestis contains a particular group of virulence factors that enable colonization in the flea and allow growth and subsistence in mammal macrophages [13]. For instance, it evolved to lose major genes for better adaptation to the flea vector. Its virulence determinants for hosts are mainly contained on 3 well-characterized plasmids (pYV/pCD1, pPla/pPCP1 and pFra/pMT), as well as in the pathogen’s chromosome [12]. Specific virulence proteins are encoded, such as Yersinia outer membrane proteins (Yops) and the protease Pla, which cause host cell death and escape from phagocytosis by macrophages through multiple sophisticated strategies [14]. In addition to the well-known virulence genes, novel virulence factors have been detected in recent years. With the introduction of genome-wide fitness profiling and mutant screening methods, more genes were identified as being involved in bacterial intracellular survival in the early stage of bubonic plague, as well as in the infection process [15,16]. Gene sequencing not only revealed the genetic information of Y. pestis, but also phylogeographic information on its evolution. An analysis of 345 core genome sequences from Y. pestis based on the National Center for Biotechnology Information (NCBI) international database and genome sequencing revealed 3,315 single-nucleotide polymorphisms (SNPs). Combining the results of phylogenetic trees and strain foci, the evolution of Y. pestis mainly occurred in Asia, including China, Mongolia, and Middle East [11].
Materials and Methods
Literature Search
The systematic review follows the Preferred Reporting Items of Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Figure 1). A literature search was performed in Medline, Embase, and Web of Science from their inception to December 2021. Firstly, articles were identified using the key terms “Yersinia pestis” or “Plague.” These words were then combined in a second search with the subject headings “Antibiotic Resistance,” “Antimicrobial Resistance,” or “Drug Resistance” to identify references related to Y. pestis antibiotic resistance.

Systematic review flow chart of the literature search. Boxes on the left: steps of the literature search, including identification, screening, and included. Boxes in the middle: n refers to the number of articles remaining after each step.
In total, 1,475 potentially related publications were identified through the database search. Twenty-four other articles were identified via a manual review of article references. As a result, 1,499 articles were screened based on the title, abstract, and content. All publications that contained content on the life cycle/transmission of Y. pestis and its antibiotic resistance were included, along with those that dealt with the clinical manifestations and diagnosis, latest treatment, and prevention of plague. Finally, 68 full-text articles were included in this systematic review. The flow chart of the literature search is shown in Figure 1.
Results
Epidemiology
As an obligate parasite, the life cycle of Y. pestis consists of rodents, transmission from fleas to animal reservoirs, and transmission between mammal hosts. Fleas feed on a rodent infected with Y. pestis, which survive within the flea gut. The moving vector then delivers the bacteria intradermally through a bite into a new mammalian host. Lagomorphs, artiodactyls, carnivores, hyracoids, insects, marsupials, and primates can be infected [4]. The movement of Y. pestis through different types of mammals promotes the distribution of plague between areas [17]. Humans can be infected with Y. pestis by flea bites, contact with infected animals, and intake of undercooked contaminated meat. Therefore, people surrounded by animals are more likely to be infected. Moreover, plague can be transmitted between people by respiratory droplets from the coughs of infected patients [18].
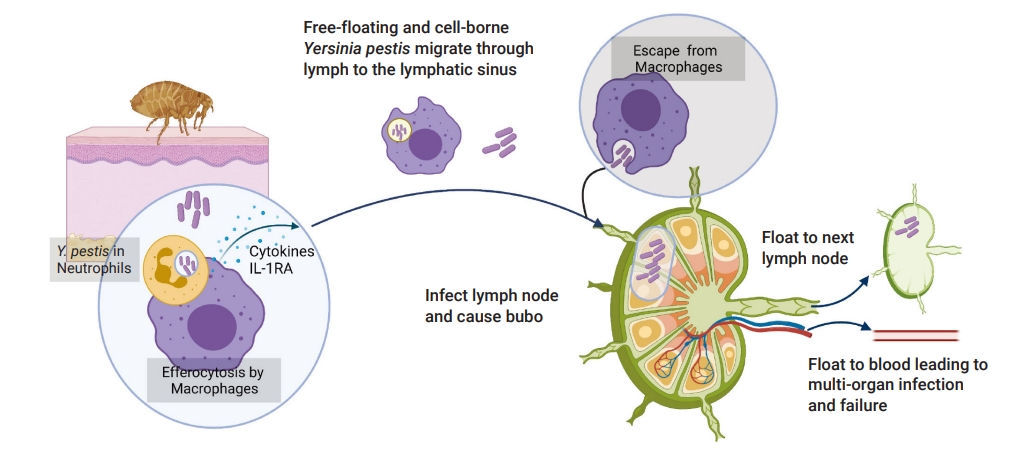
When Y. pestis enters in vivo, the infection begins (Figure 2). Firstly, it can enter neutrophils and macrophages actively or passively. It has generally been thought that Y. pestis bacteria are killed in neutrophils, while they can live on and proliferate within macrophages [19]. Nevertheless, it has been proven that Y. pestis can also survive and duplicate before the type III secretion system (T3SS) is upregulated at the body temperature of fleas [20]. After that, infected neutrophils would be ingested by macrophages in a cleaning process known as efferocytosis [12]. During the efferocytosis period, a cytokine named 1L-1RA is secreted, which blocks the inflammatory response and upregulates the T3SS [21]. This may partly explain why Y. pestis adapts to the extracellular lifestyle with antiphagocytic abilities even after escaping from macrophages. Thus, it is difficult for phagocytes to ingest the bacteria in the late stage of the infection. As the bacteria arrive at the target organs via lymphatic system, virulence factors such as Yops translocate into the host cells through T3SS, which both activates the signal-transducing pathway and promotes contact between Yops and host cells [13].
Detection and Diagnosis of Plague
A rapid diagnosis is required to ensure that plague-suspected patients receive timely medical administration to reduce the risk of mortality. Several biological diagnostic tests have been developed to identify Y. pestis. Isolation of this pathogen from clinical specimens remains the gold standard for plague diagnosis according to World Health Organization recommendations [12]. The use of selective cefsulodin-irgasan-novobiocin medium under an appropriate culture temperature are essential to isolate the bacteria [22]. Nonetheless, this method is quite time-consuming (about 3 to 5 days) and labor-intensive [12]. Therefore, automatic detection methods have been developed for faster diagnostic results. The F1 antigen has been widely adopted as the target for discriminating Y. pestis, since it is the typical immunogenic protein of the pathogen [23–25]. The F1 dipstick assay and enzyme-linked immunosorbent assay have been developed to identify the F1 antigen, and these methods have been proven practical with trained staff at local points of care [12]. However, these methods that depend on an immunological reaction have some limitations, including low sensitivity and delayed detection [26,27]. The detection results may be negative if samples are collected in the early stage of infection when massive multiplication has not occurred inside the host [27]. Other detection methods, such as phage lysis and mass spectrometry, are sometimes misleading [12,28].
Molecular genetic tests have been developed to reduce delays in detection and increase the sensitivity of diagnostics. Conventional polymerase chain reaction (PCR) targeting the caf1, pla, yopM, and inv gens can offer results within 3 to 4 hours [12]. Nonetheless, PCR-based diagnostic methods need to be performed in a laboratory with level II biosafety, technical expertise, and specialized instruments [29,30]. Thus, portable real-time quantitative PCR thermocyclers have been invented to enable medical staff to perform tests in the field without special equipment [18]. The recombinase polymerase assay and loop mediated isothermal amplification (LAMP) assay have been developed to make the tests easier and more efficient than conventional PCR [26]. A LAMP assay pair on caf1 and 3a has been described, but it was only tested in artificially spiked human blood samples [26]. Another LAMP assay targeting the caf1 gene was developed for detection of Y. pestis in plague biological samples. The sensitivity and specificity of this test were quite reliable compared with the gold standard [31]. In fact, the diagnosis of Y. pestis is generally confirmed by a combination of several detection methods. Culture-based identification offers provisional results, while molecular biology tests or antigens detection tests provide more precise results [27].
Clinical Manifestations
There are numerous specific clinical manifestations of plague depending on the route of transmission [32]. Three major types of plague are generally reported: bubonic, septicemic, and pneumonic [33]. As the most frequent form, bubonic plague is characterized by swollen regional lymph nodes and usually occurs after infected flea bites. In some cases, patients may suffer red and hot skin, headache, fever (up to 39°C), chills, malaise, and severe pain of the lymph nodes near the flea-bite region [18]. Nausea, emesis, and dizziness are less-common manifestations. Without treatment in time, Y. pestis can spread to the lungs and other organs, leading to more severe clinical symptoms [4]. Septicemic plague occurs when the blood is infected. This type may arise primarily and secondarily to bubonic plague, generally accompanied by systemic symptoms, including sudden chills, high fever, multiple organ failure, nausea, and diarrhea. Disseminated intravascular coagulation, purpura and acral cyanosis, necrosis, and hemorrhage in the skin and serosal surfaces may present later.
The most virulent and dangerous type is pneumonic plague, which results in nearly 100% mortality and is easily transmissible via airborne droplets. The incubation period is quite short, usually 1 to 3 days, after large-scale exposure to Y. pestis. This form manifests with the abrupt onset of chills, fever, weakness, headache, chest discomfort, severe coughing, and sometimes bloody or watery mucus [34].
Other forms of plague are rarely reported. Pharyngeal plague sometimes occurs when people eat uncooked meat contaminated by Y. pestis [4]. This form of plague may present with distinctive clinical symptoms, including acute pharyngitis and tonsillitis, accompanied by enlargement of the cervical lymph nodes [35]. Gastrointestinal plague is transmitted in the same way, and it manifests as vomiting, abdominal pain, diarrhea, tenesmus, and mucous stool accompanied with systemic symptoms [36]. Cutaneous plague is also described in rare cases. The infection is transmitted through flea bites, with the rapid formation of herpes and pustules [18].
Treatment
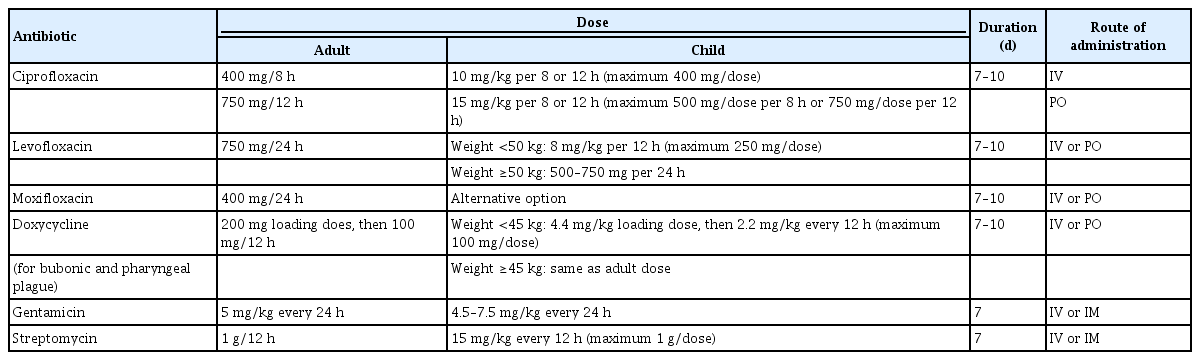
Although plague, especially the pneumonic form, can cause high and rapid mortality, the disease can be treated with early antimicrobial treatment [37]. Based on a systematic review of 762 published clinical cases from 1937 to 2019, aminoglycosides, tetracyclines, fluoroquinolones, and sulfonamides proved to be effective against plague. However, the case fatality rate of patients treated with sulfonamides was slightly higher, which is likely due to the time period, since sulfonamides were generally used from 1937 to 1949. Fluoroquinolones have been exclusively administered since the 1980s, when they were developed [38]. According to the latest recommendations on antimicrobial treatment for plague from the Centers for Disease Control and Prevention (CDC), aminoglycosides and fluoroquinolones are the mainstays of antimicrobial treatment for plague, while tetracyclines, chloramphenicol, and trimethoprim-sulfamethoxazole (TMP-SMX) could also be options for treatment depending on the age and pregnancy status of the patient [37]. Nevertheless, a study of plague in Vietnam revealed that patients treated with TMP-SMX had a longer duration of fever and some developed complications [38]. Furthermore, the World Health Organization does not recommend chloramphenicol as plague therapy considering its severe adverse effects, including reversible bone marrow suppression, aplastic anemia, and “gray baby” syndrome. Tetracyclines have few major adverse events, but they are contraindicated for pregnant women and infants [39]. Table 1 displays the first choices of treatment protocols for plague from the CDC [37]. Apart from doxycycline (for bubonic and pharyngeal plague), the other antibiotics in the table can be used to treat all forms of plague.
Prevention
Evidence shows that pneumonic and septicemic plague spread more easily from person to person because bloody sputum and aerosols mixed with Y. pestis are infectious [40]. Preventive measures comprise physical isolation, oral prophylactic antibiotics, and vaccines.
Patients are recommended to stay physically isolated for the initial 48 hours during antibiotic treatment until they improve clinically. Face masks, gloves, and eye protectors are standard precautions against respiratory droplets [41].
Close contacts (<2 m) of patients infected with pneumonic plague and individuals exposed to airborne droplets are at high risk of infection [34]. Therefore, they are suggested to receive orally administered ciprofloxacin, levofloxacin, doxycycline, and moxifloxacin as the first options for 7 days according to the CDC recommendations [37]. However, in a mass casualty setting, doxycycline, ciprofloxacin, and tetracycline are suggested as the preferred choice for postexposure prophylaxis based on the suggestions of Working Group on Civilian Biodefense. In contrast, gentamicin or streptomycin are recommended when a modest number of patients need treatment [42].
Vaccination is another precautionary method against the plague. A live attenuated vaccine was invented and applied in 1931 in Madagascar and remained in use in Russia and China [43]. In addition, a killed whole-cell vaccine was licensed and used by the United States Army. However, it was not continued since studies proved that this type of vaccine was only effective against bubonic plague without the ability to prevent pneumonic plague [34]. Most advanced research in recent years focused on molecular vaccines against pneumonic plague. The F1 and V antigens of Y. pestis were combined, since some strains do not contain the F1 antigen [44]. Animal trials showed that 2 doses of the F1/V vaccine could completely protect mice from pneumonic and bubonic plague. Nevertheless, no tests of the F1-V were conducted in humans [12]. Additionally, DNA vaccines have been developed as part of a prime-boost strategy by attenuating Y. pestis strains with genetic engineering, but none have progressed to the clinical stage [4]. An oral vaccine with live attenuated Y. pseudotuberculosis strains, designated as Yptb5, was recently developed. Although this mutant can provide complete protection against a pulmonary challenge with 5.5×103 colony-forming units (CFU) of Y. pestis, while partial protection (50% survival) against 100LD50 of Y. pestis [45].
Antibiotic Resistance
Antibiotic resistance or antimicrobial resistance (AMR) refers to the ability of microbes (including bacteria, fungi, and parasites) to resist antibiotics, making antimicrobial drugs ineffective [46]. Since antimicrobials are the primary options for plague treatment and prophylaxis, antibiotic resistance in Y. pestis increases the risks posed by this disease and makes it more challenging to control. Understanding the mechanism of AMR in Y. pestis is the key to solving this problem.
Y. pestis strains with antibiotic resistance
To date, 5 Y. pestis strains with unique AMR and multidrug resistance (MDR) were isolated and identified in Madagascar with complete genome sequencing (Table 2) [47]. In addition, 1 streptomycin-resistant strain of the bacterium was found, and the genome was recently sequenced in the Tibetan region of China [48].
Y. pestis strains 12/87, 56/13, 59/13, and S19960127 exhibited high streptomycin resistance due to a novel mechanism, namely a ribosomal protein S12 gene (rpsl) mutation. For isolates 56/13, 59/13, and 12/87, the minimum inhibitory concentrations (MICs) were over 1,024 μg/mL, sharing 1 SNP (position 215,373 in the CO92 reference genome). However, strain 12/87 was highly distinct from strains 56/13 and 59/13 based on a phylogenetic analysis. However, as the first strain discovered exhibited the novel mechanism of antibiotic resistance, S19960127 displayed quite high resistance to streptomycin (MIC=4,096 μg/mL). It shared the same SNP (215373) as those found in Madagascar [47,48].
Plasmid-mediated high-level resistance to antibiotics was identified in strains 17/95, 16/95, and IP2180H (Table 2). Strain 17/95 presented resistance to at least 8 antimicrobial agents to different degrees, including streptomycin (MIC>2,048 μg/mL), ampicillin (MIC=2,048 μg/mL), kanamycin (MIC=2,048 μg/mL), spectinomycin (MIC=2,048 μg/mL), sulfonamides (MIC=1,024 μg/mL), tetracycline (MIC=1,024 μg/mL), minocycline (MIC=512 μg/mL) and chloramphenicol (MIC=128 μg/mL). However, it was susceptible to TMP [49]. Y. pestis 16/95 was only resistant to streptomycin (MIC=1,024 μg/mL) and remained susceptible to others therapeutics or prophylactics against plague. Strain IP2180H exhibited high-level resistance to doxycycline (MIC>16 μg/mL), while it was susceptible to other antibiotics such as ampicillin, kanamycin, moxifloxacin, levofloxacin, and imipenem [50].
Apart from the above strains, some reported AMR/MDR strains, however, did not provide a genetic basis and transferability of resistance [51,52]. For instance, an MDR Y. pestis strain, MNG 3122, was isolated in Mongolia from a marmot. However, its antibiotic susceptibility profile was different from that of Y. pestis 17/95. It presented drug resistance to gentamicin, tetracycline, doxycycline, TMP-SMX, and chloramphenicol [52]. In addition, some AMR strains were discovered during 1996 to 1998 in Madagascar, including ampicillin-resistant and chloramphenicol-resistant isolates from a clinic in Mahajanga District. A tetracycline-resistant isolate from a rat and ampicillin-resistant isolate from a flea were discovered in Antananarivo District [51].
Recent studies on the detection of the strains of Y. pestis with AMR have been conducted (Table 3) [53–55]. PCR and susceptibility tests were primarily used. In the PCR method, various AMR-associated genes were targeted to determine the strains with resistance based on the resistance phenotype of Y. pestis of interest in a particular study. The results were all negative, as listed in Table 3 [53–55].
Transferability of antibiotic resistance in Y. pestis
The horizontal transfer of DNA between unrelated organisms is a significant source of variation that leads to new strains of bacterial pathogens. As discussed above, 3 isolates (17/95, 16/95, and IP2180H) presented antibiotic resistance attributed to conjugative plasmids with high rates of transferability, which could be spread to endemic and foreign plague populations, as well as other bacteria, in the same way [4]. These plasmids can be transferred horizontally along with other transferable elements, including transposons and integrons, leading to increased phenotypic diversity [56].
As a member of the A/C incompatibility group of plasmids, the antibiotic plasmid pIP1202 of Y. pestis 17/95 can transfer to Y. pestis 6/69cN and Escherichia coli K802N at frequencies of 1.5×10–2 and 1×10–2 per donor CFU, respectively. Meanwhile, pIP1202 can retransfer from E. colii to Y. pestis and E. colii at frequencies of 1.1×10–4 and 5.7×10–5, respectively. The lack of stability of pIP1202 in E. colii can be attributed to the lower frequency of its transfer from E. colii to other strains [49].
As for the self-transferable plasmid pIP1203 in Y. pestis 16/95, high-frequency transfers were observed from 16/95 to Y. pestis 6/69cN and Y. pseudotuberculosis IP32790cN (3×10–1 and 1×100 per donor CFU, respectively). Moreover, retransfer of the plasmid pIP1202 occurred from Y. pestis 6/69cN to Y. pestis 6/69cNR and Y. pseudotuberculosis IP32790cNR at frequencies of 2×10–1 and 5×10–1 per donor CFU, respectively. Plasmid pIP1203 was found to belong to the IncP group [57].
The mobile plasmid pIP2180H (a member of the IncH1group) can transfer from Y. pestis IP2180H to Y. pestis CO92 (1.5×10–1 per donor CFU), Y. pseudotuberculosis (5×10–2 per donor CFU) and E. colii (2×10–1 per donor CFU) at quite high transfer frequencies [50].
For strains carrying mutated rpsl genes, a study documented that these AMR Y. pestis isolates can be transmitted among individuals via respiratory droplets during a pneumonic plague outbreak [47]. Since bubonic plague can cause secondary pneumonic plague without appropriate treatment, an immediate surveillance system seems quite important to detect and identify patients infected with AMR strains.
Mechanisms of antibiotic resistance in Y. pestis
Plasmid-mediated antibiotic resistance
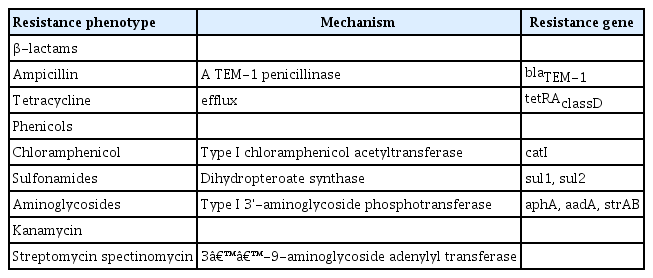
Plasmid IP1202 (182913bp) in Y. pestis 17/95 confers the ability to produce a series of proteins that inactivate antimicrobial agents. Its resistance mechanisms have constantly been revealed at the molecular level, as presented in Table 4 [42,49]. As this isolate remained susceptible to TMP, synergism did not occur between sulfonamides and TMP for plague treatment [42]. Some of the antibiotic resistance genes, including aadA, sul1, tetRA, and blaTEM-1, were located in the Tn21 transposon [58], which has been commonly found as the carrier of diverse resistance genes [59].
Streptomycin resistance in Y. pestis 16/95 was due to the plasmid pIP1203 (40 kbp), in which the strA (801 bp) and strB (834 bp) genes encoded aminoglycoside 3"-O-phosphotransferase [aph(3")-I] and a 6-O-phosphotransferase [aph(6)-I] [57]. These 2 determinants were part of the Tn5393 transposon, which is generally found in phytopathogenic bacteria, such as Snodgrassella alvi, Salmonella enterica serovar Typhimurium, and Aeromonas spp. [60–62]. The inverted terminal repeat (IR), which is frequently found at the same position in the str genes in various DNAs, was identified downstream from the strB genes. Additionally, a partial sequence of the tnpR gene of Tn5393 was located upstream from strA. The sequences IR-tnpR-strA-strB-IR were inserted in the R751backbone [42].
Y. pestis IP2180H exhibited single resistance to doxycycline and susceptibility to levofloxacin, moxifloxacin, kanamycin, ampicillin, and imipenem. An additional 171 kbp plasmid was discovered in the strain, designated pIP2180H. Its antibiotic resistance was conferred by the production of tetracycline efflux protein encoded by tetB genes located in the Tn10 transposon in the plasmid. Tn10 also carried tetR and tetC, which coded for regulators. It was also highly homological to the pB71 plasmid, a multidrug-resistant plasmid in a Salmonella enterica strain. However, the pIP2180H plasmid did not present resistance to the same antimicrobial agents as the S. enterica strain did [50].
Gene mutation-associated streptomycin resistance
As a first-line treatment option against plague, streptomycin can bind to the aminoacyl-tRNA recognition site of 16S rRNA (rrs), impairing translational proofreading and leading to the blockade of protein synthesis and subsequent cell death of sensitive bacteria [63]. Ribosomal protein S12 (rpsl) increases the stabilization of the high-order pseudoknot structure formed by rrs; therefore, amino acid substitutions in the rpsl protein influence the conserved structure of rrs and induce the failure of interactions between streptomycin and rrs, conferring high-level streptomycin resistance [48,64].
Based on whole-genome sequencing and alignment analysis with the CO92 strain, 4 isolates (56/13, 59/13, 12/87 and S19960127) were found to have an rpsl gene mutation at 128 bp, resulting in the amino acid substitution of Lys to Arg at site 43 (K43R) in the rpsl protein [48]. Subsequently, as mentioned before, this mutation disrupted streptomycin binding to rrs by altering its tertiary structure.
Mutations associated with streptomycin resistance have been consistently identified in the rpsl, rrs, and gidB genes in Mycobacterium tuberculosis (MTB) since 1993 in diverse geographic localities [65]. In contrast, this mechanism of AMR in Y. pestis was never reported until 2021, while Y. pestis strains containing mutated rpsl genes have existed since 1987 according to recent studies [47]. It is probable that detecting strains with a single method (Table 3) [53–55], such as PCR for target genes in plasmids, would reduce the accuracy of streptomycin susceptibility monitoring since it may have missed gene-mutated isolates.
The reasons for the rpsl gene mutation were not further investigated through in vivo or in vitro tests. However, a study about streptomycin resistance in MTB isolates in Nepal assumed that the inappropriate practice of streptomycin monotherapy for MTB in the region was an important factor [63]. As a first-line treatment option for plague, streptomycin is easier to access in developing countries, leading to its wide application in clinical settings, which probably caused the drug-induced gene mutation.
Two strains (17/95, S19960127) have complete genome data in the GenBank, while strains 16/95 and IP2180H only had sufficient gene annotations of specific plasmids. Information on the genes associated with AMR is listed in Table 5.
Other possible mechanisms of drug resistance
Bacterial membrane vesicles (BMVs) have recently been investigated to clarify their connection with antibiotic resistance in pathogenic and non-pathogenic bacteria. They are released by bacteria themselves. A study showed that BMVs could transform bacteria with genes encoding enzymes, resulting in their dissemination and increased AMR. Eddy et al. [66] found that the BMVs of Y. pestis contain a penicillin-binding protein activator that regulates peptidoglycan synthesis. However, detailed the mechanism of BMVs against antibiotics was not investigated in that study.
Discussion
Based on the mechanism of AMR/MDR in strains 17/95, 16/95, and IP2180H, it can be inferred that the transposons cause the transmission of antibiotic resistance genes in bacteria. Ten resistance determinants were identified in pIP1202, mostly encoded within the Tn21 transposon, resulting in MDR. In contrast, all the antibiotic resistance-associated genes in pIP1203 and pIP2180H were characterized in Tn5393 and Tn10, respectively. The 3 transposons identified in Y. pestis are typical carriers of AMR genes, which have been widely discovered in various bacteria [59]. Tn10 is a composite transposon, while Tn5393 and Tn21 are non-composite transpositions. The former type, carrying drug resistance genes, poses the most serious challenge for treatment against infectious diseases since these transposons allow bacteria to survive toxic compounds created by humans [67]. However, non-composite transposons play a very important role in the genetic evolution of bacteria and the spread of AMR due to transpositions in the transposons, which can cause the increase and decrease of bacterial virulence [59]. As a result, actions should be taken to prevent the transmission of these mobile elements among bacteria.
To date, there was not sufficient evidence confirming the reason for the rpsl gene mutation in Y. pestis with streptomycin resistance. However, cases in Madagascar that occurred in 2013 excluded streptomycin application-induced gene mutation, since sputum samples from patients surviving from plague were collected before the administration of streptomycin [47,48]. As for the cases in 1996 in Qinghai-Tibet plateau, 4 people were involved in the plague outbreak and the S19960127 strain was isolated from the necropsy organ samples from 1 patient who received streptomycin treatment for 8 days [48]. It seems that further research should be conducted in vivo or in vitro to clarify the origin of streptomycin resistance in Y. pestis. As discussed above, the frequent use of streptomycin in plague treatment may have played an important role in the emergence of this mutation. Since drugs mediate gene mutations, vaccines and antibiotics targeting novel gene points (such as DNA adenine methyltransferase) may be an inevitable choice for defeating the bacteria [68].
It should be noticed that no plague patients in Tibet who were treated with unified administration (intramuscular streptomycin, oral TMP/SMX, oral tetracycline) survived, while all cases given a similar treatment recovered in Madagascar in 2013 [47,48]. The significant difference in the treatment effects between the 2 outbreaks, on one hand, corresponded to the streptomycin suspensibility tests, in which the S19960127 strain exhibited higher streptomycin resistance than 59/13 and 56/13. On the other hand, more advanced medical conditions probably contributed to the higher cure rate of plague in the more recent outbreak. Last, it should be noted that the 3 patients infected with pneumonic plague in Tibet died quite soon after treatment (3 to 8 days) due to inappropriate medication. Not only does this fact illustrate the high mortality of pneumonic plague, but it also underscores the importance of rapid detection of antibiotic resistance of strains isolated from people in the early stage of infection. Drug susceptibility monitoring of Y. pestis isolates should not only include whole-genome sequencing to identify the well-known resistance genes, but also drug suspensibility testing based on globally accepted guidelines. The whole-genome sequencing technology should aim to offer immediate and accurate resistance information on the genetic level so that AMR/MDR strains could be identified in time, helping clinicians to initiate appropriate treatment strategies in a timely manner. Drug suspensibility testing should be conducted to discover Y. pestis strains with unknown drug resistance genes to avoid missing AMR/MDR isolates.
Conclusion
Y. pestis can acquire antibiotic resistance in nature not only via conjugative transfer of AMR plasmids from other bacteria, but also by gene point mutations. The health care system should enhance the surveillance of Y. pestis by taking measures to control transposon transmission and detect antibiotic-resistant strains with whole-genome sequencing and drug suspensibility testing. Discovering drugs targeting novel gene points is also of urgent importance considering drug-induced gene mutations.
Notes
Ethics Approval
Not applicable.
Conflicts of Interest
The authors have no conflicts of interest to declare.
Funding
None.
Availability of Data
All data generated or analyzed during this study are included in this published article. Other data may be requested through the corresponding author.
Additional Contributions
Management and Science University (Selangor, Malaysia) provided statistical support.