Articles
- Page Path
- HOME > Osong Public Health Res Perspect > Volume 5(Suppl); 2014 > Article
-
Original Article
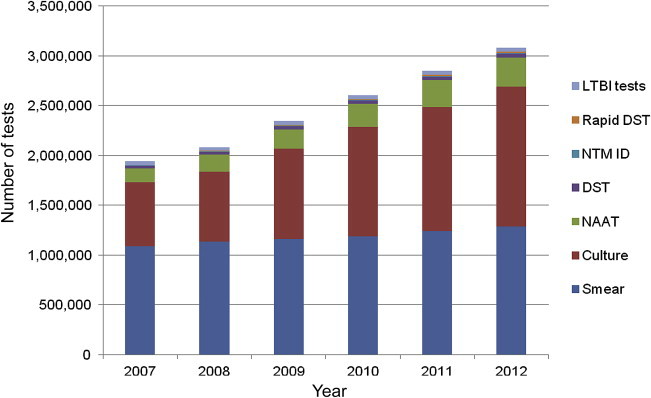
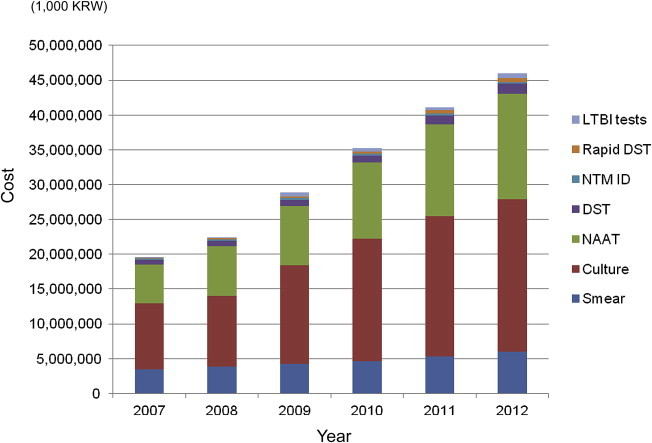
Utilization of Laboratory Tests for Tuberculosis and Mycobacterial Disease in Korea - Chang-Ki Kima, Sung Won Choia, Mi-Sun Parkb
-
Osong Public Health and Research Perspectives 2014;5(Suppl):S24-S29.
DOI: https://doi.org/10.1016/j.phrp.2014.10.008
Published online: November 4, 2014
aKorean Institute of Tuberculosis, Cheongju, Korea
bDivision of Tuberculosis and Bacterial Respiratory Infections, Korea National Institute of Health, Cheongju, Korea
- ∗Corresponding author. psoas95@gmail.com
• Received: October 15, 2014 • Revised: October 26, 2014 • Accepted: October 27, 2014
© 2014 Published by Elsevier B.V. on behalf of Korea Centers for Disease Control and Prevention.
This is an Open Access article distributed under the terms of the CC-BY-NC License (http://creativecommons.org/licenses/by-nc/3.0).
Figure & Data
References
Citations
Citations to this article as recorded by 

- Tuberculosis Surveillance and Monitoring under the National Public-Private Mix Tuberculosis Control Project in South Korea 2016–2017
Jinsoo Min, Hyung Woo Kim, Yousang Ko, Jee Youn Oh, Ji Young Kang, Joosun Lee, Young Joon Park, Sung-Soon Lee, Jae Seuk Park, Ju Sang Kim
Tuberculosis and Respiratory Diseases.2020; 83(3): 218. CrossRef - Is Tuberculosis Still the Number One Infectious Disease in Korea?
Hae-Wol Cho, Chaeshin Chu
Osong Public Health and Research Perspectives.2014; 5: S1. CrossRef


 PubReader
PubReader Cite
Cite
